P Hyundai ADM Hits Limit; NeoImmuneTech Rallies on ARS Survival[K-bio pulse]
created on 08/29/2025 10:00:55 AM
Unauthorized reproduction or distribution is illegal and subject to criminal penalties.
Pharm Edaily enforces a zero-tolerance policy and will take strict action.
Pharm Edaily enforces a zero-tolerance policy and will take strict action.
This article was released as Pharm Edaily Premium Content on 08/29/2025 10:00:55 AM
Subscribe
[Seungkwon Kim, Edaily Reporter] On Aug. 28, South Korea’s pharmaceutical, biotech and health care stocks rallied, led by companies that either secured presentations at major international conferences or posted encouraging clinical outcomes.
Hyundai ADM Hits Limit on U.S. Cancer Conference News; Hyundai Bioscience Jumps
Hyundai Bioscience and Hyundai ADM jumped together after news that an abstract was accepted for presentation at a top-tier conference. NeoImmuneTech advanced on efficacy signals from a primate study, and Protia rose on expectations of continued revenue growth after a recent swing to profit.
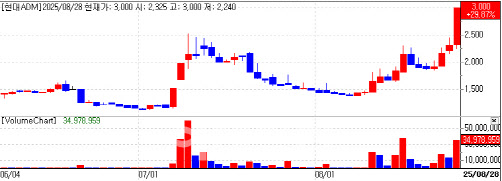
Hyundai ADM surged to the exchange’s daily limit early and closed up 29.8% at 3,000 won, according to MPDOCTOR by KG Zeroin (formerly Market Point). The gain followed word that the company’s abstract on its drug candidate Penetrium was accepted for a poster at AACR 2025, a tri-society meeting jointly hosted by the American Association for Cancer Research (AACR), the U.S. National Cancer Institute (NCI) and the European Organisation for Research and Treatment of Cancer (EORTC).
The accepted abstract, titled “Disrupting the oncobiosphere: CAF-targeting therapy with Penetrium reverses pseudo-resistance in tumors,” posits that structural barriers in the tumor microenvironment stiffened extracellular matrix and related factors rather than genetic resistance alone, underlie treatment failure. Penetrium is a reformulation of the anthelmintic niclosamide designed to improve bioavailability. By selectively dismantling the rigidified tumor microenvironment while sparing normal fibroblasts, the candidate aims to reopen tumors to existing chemotherapies and immunotherapies, potentially benefiting patients with metastatic and treatment resistant disease. “This abstract acceptance internationally recognizes a paradigm that could address fundamental limits of current cancer therapy,” CEO Cho Won-dong said, adding that Hyundai ADM will work with parent Hyundai Bioscience to move Penetrium quickly into global development and commercialization.
Hyundai Bioscience also climbed, finishing up 25% at 6,660 won. Beyond the affiliate’s abstract news, investors cited expectations for TPOXX (tecovirimat) supply in smallpox and mpox, alongside the company’s pipeline momentum. The timing drew added attention: Shortly after Nature Communications published a clinical paper on the company’s broad spectrum antiviral candidate Xafty (CP COV03), Hyundai Bioscience said it received an official invitation from the U.S. Rapid Response Partnership Vehicle (RRPV).
Recently in Magok, Seoul, Hyundai Bioscience President Bae Byung-joon met Naoki Shinahora, CEO of Japan Biotechno Pharma Co., Ltd. (JBP), to discuss securing Korean rights and building a supply chain for TPOXX. JBP holds Japanese commercialization rights to TPOXX developed by U.S.-based SIGA Technologies and supplies it to Japan’s government.
Developed under the U.S. Biomedical Advanced Research and Development Authority’s Project BioShield, TPOXX is an approved, targeted medical countermeasure in the United States, Europe and Japan and is stockpiled by governments. South Korea is also weighing procurement as part of biodefense and pandemic preparedness. The talks underscore that Xafty (pipeline) and TPOXX (approved) are distinct strategic assets: TPOXX can be supplied immediately to government stockpile markets, while Xafty advances as a broad antiviral candidate. Hyundai Bioscience said the discussions reflect an expanding partner network spanning SIGA Technologies in the United States, JBP in Japan and the Drugs for Neglected Diseases initiative (DNDi).
NeoImmuneTech, Protia Surge...Why
NeoImmuneTech rose 17% to 1,096 won after reporting survival benefits in a nonhuman primate model of acute radiation syndrome (ARS) for its T cell amplifier NT I7. ARS is caused by short term, high dose radiation exposure that can devastate the immune system and bone marrow. Under the U.S. Food and Drug Administration’s “Animal Rule,” NT I7 is being developed as a lymphocyte restoring ARS therapy, a pathway that allows approval based on adequate animal data when human trials are unethical.
According to the company, the high dose NT I7 group achieved a 60 day survival rate that was 43 percentage points higher than the irradiation only placebo control, a magnitude that is similar to or above the 38 43 percentage point improvements reported for FDA approved ARS therapies. Surviving animals also showed rapid T cell recovery, another Animal Rule criterion. Building on its first primate study, NeoImmuneTech said it will consult the FDA and proceed to a follow on study, again tracking 60 day survival and lymphocyte counts. “If NT I7 secures FDA approval as an ARS therapy, partnership and market opportunities across multiple immunologic unmet needs could expand materially,” a company official said, noting NT I7’s previously reported anti tumor activity in combination with CAR T therapies.
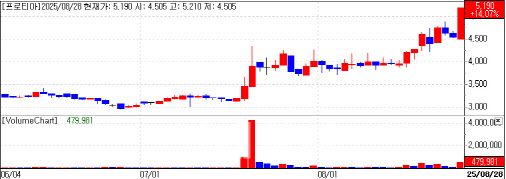
Protia climbed 14% to 5,190 won. The company’s results have been trending higher: First half revenue came to 6.54 billion won, up 41.5% year over year, and operating profit rose to 1.46 billion won from 400 million won a year earlier. Protia expects strong second half growth centered on allergy diagnostics, as its premium “PROTIA Allergy Q 192D” panel covering 176 allergens, including drug and chemical antigens enters full scale supply. The company also anticipates sharply higher demand for veterinary diagnostics in China, new sales of veterinary allergy tests in Brazil and a first launch in India of an antibiotic susceptibility test that uses an electrical capacitance measurement method.
Hyundai ADM Hits Limit on U.S. Cancer Conference News; Hyundai Bioscience Jumps
Hyundai Bioscience and Hyundai ADM jumped together after news that an abstract was accepted for presentation at a top-tier conference. NeoImmuneTech advanced on efficacy signals from a primate study, and Protia rose on expectations of continued revenue growth after a recent swing to profit.
Hyundai ADM surged to the exchange’s daily limit early and closed up 29.8% at 3,000 won, according to MPDOCTOR by KG Zeroin (formerly Market Point). The gain followed word that the company’s abstract on its drug candidate Penetrium was accepted for a poster at AACR 2025, a tri-society meeting jointly hosted by the American Association for Cancer Research (AACR), the U.S. National Cancer Institute (NCI) and the European Organisation for Research and Treatment of Cancer (EORTC).
|
Hyundai Bioscience also climbed, finishing up 25% at 6,660 won. Beyond the affiliate’s abstract news, investors cited expectations for TPOXX (tecovirimat) supply in smallpox and mpox, alongside the company’s pipeline momentum. The timing drew added attention: Shortly after Nature Communications published a clinical paper on the company’s broad spectrum antiviral candidate Xafty (CP COV03), Hyundai Bioscience said it received an official invitation from the U.S. Rapid Response Partnership Vehicle (RRPV).
Recently in Magok, Seoul, Hyundai Bioscience President Bae Byung-joon met Naoki Shinahora, CEO of Japan Biotechno Pharma Co., Ltd. (JBP), to discuss securing Korean rights and building a supply chain for TPOXX. JBP holds Japanese commercialization rights to TPOXX developed by U.S.-based SIGA Technologies and supplies it to Japan’s government.
Developed under the U.S. Biomedical Advanced Research and Development Authority’s Project BioShield, TPOXX is an approved, targeted medical countermeasure in the United States, Europe and Japan and is stockpiled by governments. South Korea is also weighing procurement as part of biodefense and pandemic preparedness. The talks underscore that Xafty (pipeline) and TPOXX (approved) are distinct strategic assets: TPOXX can be supplied immediately to government stockpile markets, while Xafty advances as a broad antiviral candidate. Hyundai Bioscience said the discussions reflect an expanding partner network spanning SIGA Technologies in the United States, JBP in Japan and the Drugs for Neglected Diseases initiative (DNDi).
NeoImmuneTech, Protia Surge...Why
NeoImmuneTech rose 17% to 1,096 won after reporting survival benefits in a nonhuman primate model of acute radiation syndrome (ARS) for its T cell amplifier NT I7. ARS is caused by short term, high dose radiation exposure that can devastate the immune system and bone marrow. Under the U.S. Food and Drug Administration’s “Animal Rule,” NT I7 is being developed as a lymphocyte restoring ARS therapy, a pathway that allows approval based on adequate animal data when human trials are unethical.
|
Protia climbed 14% to 5,190 won. The company’s results have been trending higher: First half revenue came to 6.54 billion won, up 41.5% year over year, and operating profit rose to 1.46 billion won from 400 million won a year earlier. Protia expects strong second half growth centered on allergy diagnostics, as its premium “PROTIA Allergy Q 192D” panel covering 176 allergens, including drug and chemical antigens enters full scale supply. The company also anticipates sharply higher demand for veterinary diagnostics in China, new sales of veterinary allergy tests in Brazil and a first launch in India of an antibiotic susceptibility test that uses an electrical capacitance measurement method.
김승권 peace@



![Dongwoon Jumps 8% on Saliva Test...Biosolution Looks to US [K-bio pulse]](https://image.edaily.co.kr/images/vision/files/NP/S/2026/05/PS26050400109b.jpg)

![로킷헬스케어 1.9조 수주 계약이라더니…구속력 無·매출 전환율 0.8%[only 이데일리]](https://image.edaily.co.kr/images/vision/files/NP/S/2026/05/PS26050400118b.jpg)
![지놈앤컴퍼니, 임상 본격화 기대감에 급등…숨고르기 들어간 바이젠셀[바이오 맥짚기]](https://image.edaily.co.kr/images/vision/files/NP/S/2026/04/PS26043000447b.jpg)
